Regression of dengue fever caused by infection of male Aedes aegypti mosquitoes with the Wolbachia bacterium
Summary for general audiences
The Good, the Bad, and the Ugly
This trilogy perfectly applies to the trio of bacteria, mosquito, and virus responsible for dengue fever.
The mosquito (the bad) bites a human and can transmit the virus (the ugly): the human becomes ill.
The Wolbachia bacterium (the good) infects the mosquito, blocking its reproduction and the production of the virus: the human is not affected.
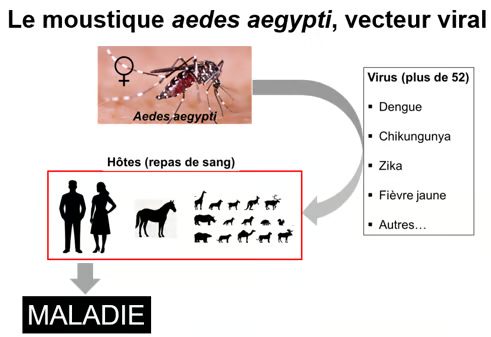
Figure. Humans are the primary host of the dengue virus, which also affects other mammals. A single bite from a female mosquito infected with the virus is sufficient to transmit the disease. For 2 to 10 days following infection, the blood of an infected person contains a high concentration of viral particles. When a female mosquito bites an infected person to feed on their blood, the virus spreads to the insect, particularly to its salivary glands. During its next bite, the mosquito injects its infectious saliva into the blood of its victim, thus spreading the disease. The Aedes aegypti mosquito is therefore the vector of many viral diseases—not just dengue—as shown in the figure.
A Brief History of the disease
Dengue is a viral disease transmitted by mosquitoes, which were its ancestral hosts. Non-human primates (macaques and Presbytis monkeys) were the first vertebrate hosts of the forest cycle that gave rise to modern dengue. The Aedes aegypti mosquito has not been the original vector. It developed as a specialized form for humans in West Africa thousands of years ago and only became the dominant urban vector after the virus had already spread from the forest cycle. Scientists estimate that transmission to humans occurred approximately 500 to 2,000 years ago (estimates vary slightly depending on the serotype and the study), once human populations in Asia and Africa had become large and dense enough to allow continuous human-to-mosquito and mosquito-to-human transmission (https://www.nature.com/scitable/topicpage/dengue-viruses-22400925/).
An article entitled
"Suppression of dengue by Wolbachia-infected male mosquitoes" by Lim et al., was recently published in the New England Journal of Medicine on March 26, 2026.
This study is based on the fact that male Aedes aegypti mosquitoes infected with the Wolbachia pipientis bacterium do not produce offspring after mating with females. This was a randomized controlled trial conducted in an urban setting in Singapore over a 24-month period, involving an estimated population of approximately 724,000. The city was divided into eight treatment zones (where Wolbachia-infected male mosquitoes were released twice weekly, at a rate of approximately one to six mosquitoes per resident per week) and seven control zones (without mosquito releases), spaced sufficiently far apart. Laboratory-confirmed symptomatic dengue cases (all severity levels and serotypes combined) were assessed using national surveillance data (RT-qPCR, NS1 viral antigen, IgM antibodies, etc.).
The figure below shows the evolution of the female mosquito population before and after the intervention.
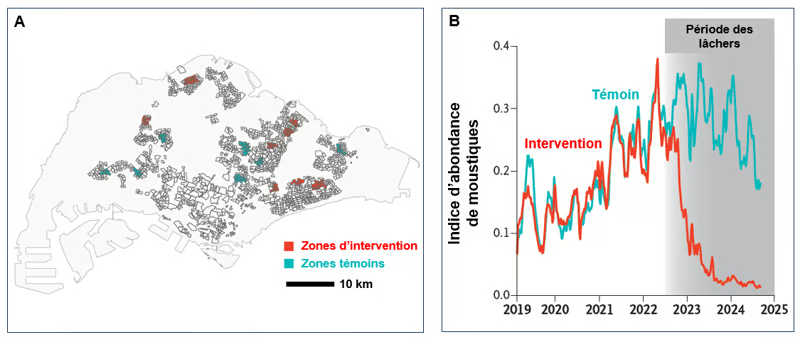
Figure. Population of Aedes aegypti mosquitoes in randomized traps. A. Map of Singapore. Spatial visualization of intervention (red) and control (turquoise) areas. B. Weekly abundance index of adult Aedes aegypti mosquitoes, measured at the trap area before (control turquoise line) and during (intervention red line) the release of Wolbachia-infected male mosquitoes (release period in gray). The index is calculated by dividing the total number of adult female Aedes aegypti mosquitoes captured in functional traps by the number of functional traps (Adapted from Lim et al. NEJM 2026).
Weekly mosquito abundance indices before the intervention were similar (approximately 0.18–0.19). Starting 3 months after the release of Wolbachia-infected male mosquitoes (and continuing up to 24 months), the index dropped sharply in the intervention areas to 0.041, compared to 0.277 in the control areas, indicating a strong and sustained suppression of the wild strain of Aedes aegypti. Furthermore, while the rate of residents testing positive for dengue in the control areas was 21–23% throughout the study, this rate fell to 6–7% among residents of the treated areas, demonstrating a reduction in the risk of dengue infection of approximately 71–72%.
It should also be noted that Wolbachia strongly inhibits the replication and transmission of the dengue virus in the A. aegypti mosquito, further contributing to its beneficial effect on disease transmission (see below).
To learn more
Dengue fever
This viral disease represents a major and growing public health problem in urban and suburban tropical areas. It is estimated that 400 million people are affected each year, and more than half of the world's population is at risk of contracting the disease (https://www.pasteur.fr/fr/centre-medical/fiches-maladies/dengue). Its mild form resembles a flu-like syndrome with joint pain. Its most severe form manifests as hemorrhagic fever with an extreme drop in blood pressure and hypovolemic shock that develops suddenly, sometimes just a few days after the fever has subsided. These symptoms constitute a medical emergency, which can precede multiple organ failure and death (Khan et al. J Infect Public Heath 2023; Wang et al. J Microbiol Immunol Infect 2020). Regarding dengue vaccines, their effectiveness is generally greater against severe forms than against mild forms, and better in people who have already been exposed to the disease. No vaccine offers 100% protection, and the current consensus is that complete control of the disease will require other approaches (Boscardin. Curr Top Microbiol Immunol 2026 ; Parra-Gonzalez et al. Front Cell Infect Microbiol 2025).
The dengue virus and its vector, the Aedes aegypti mosquito
The dengue virus, whose scientific name since 2022 has been Orthoflavivirus denguei (DENV), is an enveloped, single-stranded, positive-sense RNA virus belonging to the genus Orthoflavivirus. Five different variants of this virus are known, designated DENV-1 to DENV-5 (Murugesan & Manoharan. Emerging and Reemerging Viral Pathogens. Chapter 16. 2020). This virus is primarily transmitted by Aedes aegypti mosquitoes, which act as vectors. Humans are the main host of the virus, which also affects other mammals. A single bite from a female mosquito is enough to spread the infection if she is carrying the virus. For 2 to 10 days following infection, the blood of an infected person contains a high concentration of viral particles. A female mosquito that feeds on this blood during a bite then spreads the virus to the cells lining her gut. Over the following days, the virus spreads to the mosquito's salivary glands and is released in her saliva. During her next bite, the female injects this infectious saliva into her victim's bloodstream, thus spreading the disease. Furthermore, the dengue virus can persist in mosquito populations through transovarial transmission, in which mosquitoes infected with the virus transfer it to their eggs (Vasilakis et al. Nat Rev Microbiol 2011). The virus does not appear to have any adverse effect on the mosquito, which remains infected for life.
Ancestral origins: first, the forest cycle
The four dengue serotypes that infect humans today (DENV-1 to DENV-4) evolved independently from ancestral forest strains. These ancestral viruses circulated in a non-human primate-mosquito cycle in the forests of Southeast Asia (and separately in West Africa for some strains). The vertebrate hosts were monkeys (e.g., species of the genera Macaque and Presbytis), and the vectors were arboreal (canopy-dwelling) mosquitoes of the genus Aedes—not Aedes aegypti, as is the case today. Sylvan strains of DENV are still found in these forest cycles and form the basis of the phylogenetic tree for the human serotypes. This confirms that transmission to humans occurred from the primate-mosquito system (Wang et al. J Virol 2000 ; Vasilakis et al. Nat Rev Microbiol 2011).
Scientists estimate that transmission to humans occurred around 500 to 2,000 years ago (estimates vary slightly depending on the serotype and the study), once human populations in Asia and Africa became large and dense enough to allow continuous human-to-mosquito and mosquito-to-human transmission (https://www.nature.com/scitable/topicpage/dengue-viruses-22400925/).
Mosquito control
Traditional mosquito control measures (mosquito nets, personal protective equipment, or pesticides, in particular) have a limited or questionable ecological impact. A biological method, in use for several years, is the control of the Aedes aegypti mosquito population through X-ray sterilization or the use of the bacterium Wolbachia pipientis (Werren. Annu Rev Entomol 1997 ; Flores & O'Neill. Nat Rev Microbiol 2018 ; Werren et al. Nat Rev Microbiol 2008).
Two biological techniques for controlling mosquito populations should be considered here:
The sterilization technique using irradiation
This technique uses X-ray irradiation to sterilize male mosquitoes, which are then released in large numbers to mate with wild females, thus preventing the hatching of their eggs. This results in a decline in the wild population over time and with successive releases (Zhang et al. BMC Biol 2023).
The sterile insect technique through cytoplasmic incompatibility
In this technique, mosquitoes are reared in the laboratory in an environment containing the Wolbachia bacterium. A very interesting point to note here is that by infecting Aedes aegypti mosquitoes (males and females), the Wolbachia bacterium not only sterilizes the males, as we have just seen above, but also inhibits the replication of the dengue virus and other viruses that these insects can carry in both sexes (Flores & O'Neill. Nat Rev Microbiol 2018).
Two strategies can be used:
i) Only males are released, thus suppressing the wild population. When a male mosquito infected with Wolbachia mates with an uninfected wild female, its eggs do not hatch. The effect is similar to that of the previous technique, and over time, with repeated releases, the wild population decreases.
ii) Both sexes are infected with Wolbachia and released into the environment, thus replacing the wild mosquito population.
In both cases, the result is a situation in which viral replication, and therefore disease transmission, is reduced.
Trials conducted in recent years in various tropical countries, including New Caledonia (Nouméa and other locations https://www.worldmosquitoprogram.org/wmp-en-nouvelle-caledonie), have shown that this intervention significantly reduces the risk of dengue (70%), but the protective efficacy of this intervention varies according to geographical and human contexts (Zhang et al. BMC Biol 2023; Pinto et al. Plos Negl Trop Dis 2021; Utari et al. N Engl J Med 2021).
The Wolbachia bacterium and the mechanism of cytoplasmic incompatibility
In mosquito testes, the bacterium Wolbachia alters developing sperm at the level of the paternal chromatin/nucleoprotein complex. Mature sperm typically contain little or no Wolbachia bacteria, and the alteration therefore constitutes a lasting mark on the sperm before fertilization. This alteration disrupts the normal behavior of paternal chromosomes after fertilization, often causing problems with DNA replication, chromatin condensation, histone-protamine transition, mitotic synchronization, or chromosome segregation in the early embryo. This results in embryonic developmental arrest. For more details on the mechanism of cytoplasmic incompatibility, see the following articles (Chen et al. Genes 2020 ; Hochstrasser. Annu Rev Microbiol 2022 ; Kaur et al. Plos Biol 2022 ; Zabalou et al. PNAS 2004; Sicard et al. Curr Opin Insect Sci 2019).
The Wolbachia bacterium inhibits the replication and transmission of the dengue virus in A. aegypti
Not only does Wolbachia sterilize male mosquitoes, but this bacterium also inhibits the replication and transmission of dengue virus (DENV) in the Aedes aegypti mosquito through multiple, combined mechanisms. These mechanisms are not yet fully understood, and different Wolbachia strains (including the wAlbB strain used in the Singapore suppression trial analyzed here) exhibit varying efficacies. However, the overall effect is robust: infected mosquitoes show significantly lower rates of infection, DENV replication and dissemination to salivary glands, as well as transmission to humans. This blockage requires the presence of both the bacterium and the virus in the same cell, and it is more pronounced when the Wolbachia bacterial load is high in key tissues such as the mosquito's gut and salivary glands. It primarily affects positive-sense single-stranded RNA viruses, such as DENV, which use cytoplasmic membrane replication compartments for their replication (Hussain et al. iScience 2023; Lu et al. Front Microbiol 2020; Bian et al. Plos Pathog 2010; Mushtaq et al. Front Immunol 2024).
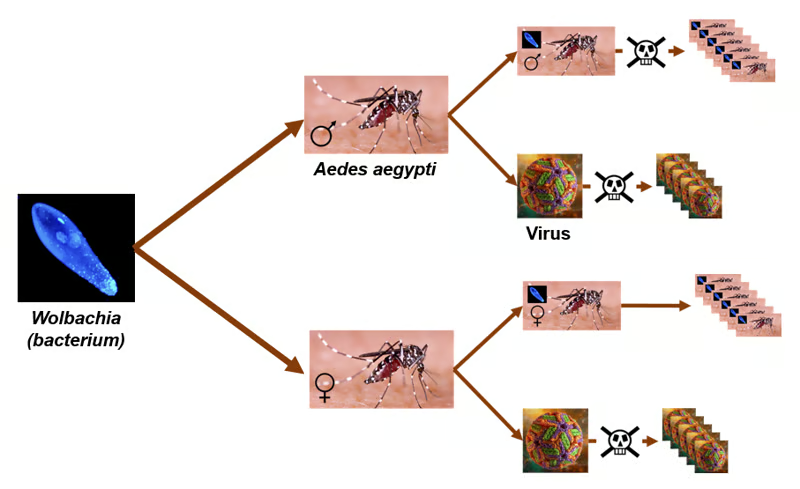
Figure. The effects of Wolbachia on the A. aegypti mosquito. Top of the figure: when a male mosquito mates with an uninfected female, the embryo is not viable (cytoplasmic incompatibility) and offspring reproduction is blocked (wild population suppression strategy). Bottom of the figure: when an infected male mosquito mates with an infected female, the cytoplasmic incompatibility is overcome and offspring reproduction is ensured (wild population replacement strategy). In both cases, DENV production is also significantly reduced.
Methods and experiments
This was a geographically randomized controlled trial conducted in densely populated urban Singapore from 2022 to 2024. Researchers divided the 12 km² city into 15 high-rise social housing zones (estimated population of approximately 724,000 residents), each about 0.8 km² in size, separated by sufficient distances to limit mosquito spread. Eight zones (393,236 residents) received the intervention; seven zones (331,192 residents) served as controls, with no mosquito releases.
Male Aedes aegypti mosquitoes (locally adapted strain) infected with Wolbachia bacteria (wAlbB strain) were released twice weekly (approximately 1 to 6 per resident per week). The releases used a local strain exhibiting complete cytoplasmic incompatibility.
Main evaluation criterion
Symptomatic dengue cases were laboratory-confirmed (all severity levels and serotypes combined) and assessed using national surveillance data (RT-qPCR, NS1 antigen, IgM, etc.). Exposure to the intervention was based on residential group (area) and duration (0, 3, 6, 9, 12 months and longer). Protective efficacy was calculated using a weighted logistic regression comparing the probabilities of exposure in dengue-positive and dengue-negative cases.
Entomological monitoring
The weekly abundance index of adult female Aedes aegypti mosquitoes was calculated using intervention and control traps before and during releases of Wolbachia-infected male mosquitoes. The index was calculated by dividing the total number of adult female Aedes aegypti mosquitoes captured in functional traps by the number of functional traps. The trial was conducted in parallel with standard integrated vector control in both groups and was registered (ClinicalTrials.gov NCT05505682).
Results
Mosquito Population Suppression
The baseline weekly mosquito abundance index was similar (approximately 0.18–0.19) in both types of areas (intervention and control). Starting 3 months post-release (and continuing up to 24 months), this index dropped sharply in the intervention areas to 0.041, compared to 0.277 in the control areas, indicating strong and sustained suppression of the wild strain of Aedes aegypti.
Dengue Incidence Reduction
In the intention-to-treat analysis (after ≥ 6 months of exposure), 6% of tests (354/5,722) were positive for dengue in the intervention areas, compared to 21% (1,519/7,080) in the control areas. Protective efficacy was 71–72% over exposure periods of 3, 6, and more than 12 months. These results were confirmed across all subgroups (age, sex, year) and remained robust to sensitivity analyses (e.g., excluding IgM-only tests). Dengue virus type 3 was predominant. The intervention reduced both vector density and the risk of dengue in humans in a real-world urban setting.
Conclusions and Implications
The authors conclude that repeated releases of Wolbachia pipientis-infected and sterilized male Aedes aegypti mosquitoes effectively suppressed mosquito populations and reduced the risk of dengue infection by approximately 71–72% among residents with prolonged exposure. This approach can complement existing vector control and vaccination strategies and contribute to reducing, or even eliminating, the transmission of dengue and other diseases transmitted by Aedes mosquitoes (e.g., those carrying Zika and Chikungunya viruses). Public acceptance was high in previous pilot trials, and the method appears applicable on a large scale in dense urban environments. Overall, this large-scale randomized trial, one of the first rigorous evaluations of releases of exclusively Wolbachia-infected male mosquitoes for dengue control, provides strong evidence for the effectiveness of this biological suppression method.
New findings in this article
This article provides the strongest randomized evidence to date that cytoplasmic incompatibility (in infected males) due to Wolbachia infection can durably suppress Aedes aegypti populations and significantly reduce the risk of dengue in a challenging, high-impact tropical urban environment. It advances the field by validating mosquito population suppression (rather than replacement with infected males and females) as a viable and potentially complementary tool that does not require the permanent establishment of Wolbachia in the wild. The design and scale of this study make it a benchmark for operational vector control policy. The results obtained allow this approach to be extended to similar contexts and to other diseases transmitted by Aedes mosquitoes.