Could tanycyte degeneration contribute to the pathology of Alzheimer's disease?
Summary for general audiences
A French study, published on February 1, 2026, by
Sauvé et al., Tanycytic degeneration impairs tau clearance and contributes to Alzheimer’s disease pathology, Cell Press Blue 2026, has just highlighted a new cellular player that could contribute to the pathology of Alzheimer's disease, tanycytes.
I have already discussed Alzheimer's disease on this site in a previous article (see here).
The generally accepted pathological characteristics of Alzheimer's disease are the accumulation and aggregation in the brain of at least two abnormal and toxic proteins: beta-amyloid protein, which forms amyloid plaques in brain tissue, and tau protein, which forms protein tangles within neurons (see here). Over the past two decades, therapies targeting the Tau protein have generated considerable interest as potential treatments for Alzheimer's disease (Bloom, JAMA Neurol 2014).
For the body, it is imperative to get rid of abnormal and toxic proteins
The concentration of Tau protein is increased in the cerebrospinal fluid (a fluid that irrigates the brain) of Alzheimer's patients, where it is present in abnormal and toxic forms. In humans, cerebrospinal fluid is considered an intermediate stage in the elimination of waste products from the brain, including Tau protein. Initially, this protein is extracted from brain tissue and directed to the cerebrospinal fluid, probably via the glymphatic system. This system is a pathway for waste removal in the brain. Subsequently, the Tau protein present in the cerebrospinal fluid is transported into the bloodstream for elimination (Grundke-Iqbal et al., Proc Natl Acad Sci U S A. 1986 ; Braak H and Braak E., Acta Neuropathol. 1991 ; Medeiros et al., CNS Neurosci Ther. 2010).
However, the details of this transfer remained incomplete. It is precisely this point that the authors of the study presented here focused on.
Their main discovery is twofold:
i) the transport of the Tau protein from the brain to the blood is carried out in the hypothalamus by cells called tanycytes
ii) tanycytes are severely damaged in the brains of Alzheimer's patients, and the elimination of the Tau protein no longer occurs
See the figure below
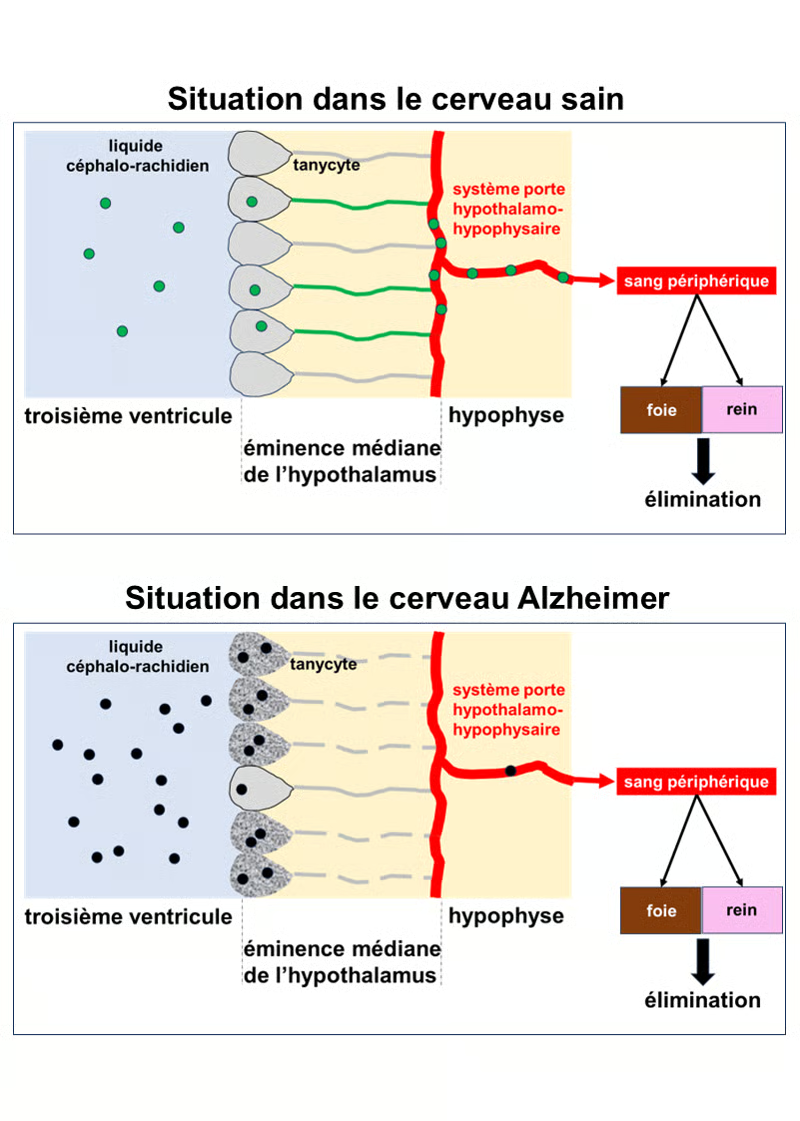
Study Results
Top section: Situation in the healthy brain. Normal Tau protein, represented by green circles, is present in the cerebrospinal fluid (light blue). It is taken up by tanycytes in the hypothalamus, which line the third ventricle of the brain. The tanycytes transport the Tau protein into the bloodstream via the pituitary portal system. This protein is then eliminated from the body by the liver and kidneys.
Bottom section: Situation in the Alzheimer's brain. The Tau protein is abnormal and toxic. It is represented by black circles. In the Alzheimer's brain, the tanycytes are degraded, and their extensions, which connect to the pituitary portal system, are fragmented and therefore non-functional (gray dotted lines). They can incorporate the Tau protein, but cannot export it into the bloodstream. It accumulates in the cerebrospinal fluid and the brain, continuing its toxic action on neurons.
Can tanycytes be repaired to stop Alzheimer's disease?
This is impossible today (March 2026), but this strategy remains a goal for researchers. By finding ways to preserve the health and integrity of tanycytes, it might be possible to maintain the brain's system for eliminating Tau protein (and others) in a functioning state, which could significantly slow the progression of the disease.
To learn more
The Tau protein
Tau protein is a natively unfolded, highly soluble, and thermostable protein encoded by the MAPT (microtubule-associated protein tau) gene on chromosome 17. The adult human brain expresses six isoforms of this protein (352 to 441 amino acids) generated by alternative splicing: combinations of 0 to 2 N-terminal insertions (0N/1N/2N) and 3 or 4 microtubule-binding repeats (3R/4R). The longest isoform (2N4R, 441 amino acids) is the most common.
In neurons, this protein is associated with microtubules (tubulin polymers), which are the constituent fibers of the cytoskeleton (a kind of railway track along which molecules, proteins, and organelles necessary for the life and function of neurons travel) at the level of axons and dendrites. One of the main functions of the Tau protein is to stabilize microtubules. To do this, it binds to microtubules (with an affinity of approximately 100 nM at neuronal concentrations of about 2 μM), promotes tubulin polymerization, stabilizes the microtubule network, and plays a role in axonal transport, neuronal morphology, and intracellular trafficking. The Tau protein is a phosphoprotein.
Here is a list of references on this protein: Wang et al. Nature Reviews Neuroscience 2016; Chong et al. Cell and MolNeurobiol 2018; Shen et al. Cells 2025; Grundke-Iqbal et al., Proc Natl Acad Sci U S A. 1986; Hong et al. Biomolecules 2025; Wegmann et al. Curr Opin Neurobiol 2021.
In the normal brain, its phosphorylation is regulated at a low level (2 to 3 moles of phosphate per mole of tau) by a balance between kinases (e.g., GSK-3β, CDK5) and phosphatases (e.g., PP2A). This allows for dynamic attachment and detachment without disrupting function. Normal tau protein remains monomeric and soluble and does not aggregate. The attachment of these phosphate groups is tightly controlled because it critically impacts microtubule stability.
In the Alzheimer brain, the Tau protein undergoes abnormal hyperphosphorylation (3 to 4 times greater than normal, reaching 6 to 8 phosphate molecules per mole of Tau, resulting in approximately 39 to 45 phosphorylated sites in the hyperphosphorylated Tau protein). Key sites include Ser202/Thr205, Thr231, Ser262, Ser396/404, etc. This phenomenon is often associated with reduced phosphatase activity (e.g., PP2A) and kinase overactivation. The hyperphosphorylated Tau protein detaches from microtubules, inhibits their assembly, and causes their disassembly. It sequesters normal Tau protein and other MAPs (microtubule-associated proteins, MAP1/MAP2) in the cytosol.
Once detached from microtubules, the Tau protein misfolds, forming oligomers → dimers → paired helical filaments or straight filaments → insoluble neurofibrillary tangles within neurons (at the soma and dendrites). All six isoforms are present in these neurofibrillary tangles. Further modifications, such as acetylation (e.g., Lys280), truncation (e.g., at Asp421 or Glu391 by caspases), and ubiquitination, promote aggregation. The oligomeric forms are highly toxic and spread similarly to prions (seeding).
Hyperphosphorylated Tau protein migrates from axons to the central compartment and dendrites. Neurofibrillary tangles damage the protein's transport pathways, inducing inflammation, synaptic dysfunction, and neuronal death. The presence of neurofibrillary tangles is strongly correlated with cognitive decline and disease severity—often more so than amyloid plaques—and appears decades before symptoms.
Thus, in a healthy brain, the tau protein is essential and protective when properly regulated. In Alzheimer's disease, hyperphosphorylation transforms it from an ally into an enemy: it loses its stabilizing role and acquires toxic properties that lead to neuronal death and the progression of symptoms. Tau protein is now a major therapeutic target (e.g., anti-Tau antibodies, kinase inhibitors), and biomarkers such as phosphorylated Tau protein in cerebrospinal fluid or Tau PET imaging allow, in many cases, for better monitoring of disease progression, much better than with beta-amyloid protein alone.
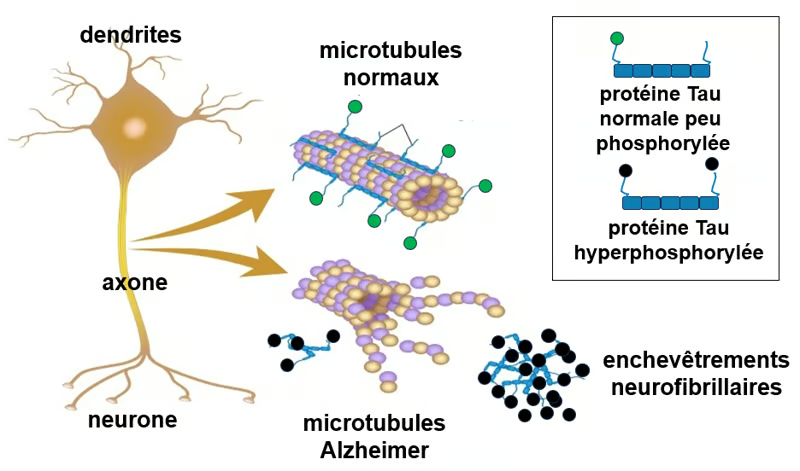
Figure. Tau protein and microtubules. One of the main functions of the Tau protein is to modulate the stability of axonal microtubules. The axons and dendrites of neurons are very elongated parts of the cell that require microtubules for structural and functional support. Like many other proteins, the Tau protein is subject to modifications, notably through the binding of phosphate groups, which alter its function. In the healthy adult human brain, the Tau protein undergoes physiological (normal) phosphorylation at a limited number of sites, on average 2 to 3 phosphate groups per molecule (Tau protein symbolized, bearing a single green circle). In contrast, in the Alzheimer's brain, the pathological Tau protein exhibits hyperphosphorylation (often of 3 to more than 10 phosphate groups per molecule) at numerous additional sites (symbolized by two black circles), leading to a decreased affinity for microtubules, microtubule disorganization, and aggregation into neurofibrillary tangles, resulting in neuronal dysfunction and degeneration. Source: adapted from Alila Medical Media, Shutterstock.
Hypothalamus and tanycytes
The hypothalamus is a small structure in the vertebrate brain containing several nuclei with diverse functions. In humans, its size is comparable to that of an almond. One of its functions is to provide the link between the central nervous system and the endocrine system via the pituitary gland. It thus controls body temperature, hunger, important aspects of parenting and maternal attachment behaviors, thirst, fatigue, sleep, circadian rhythms, and is important in certain social behaviors, such as sexual and aggressive behaviors (Hypothalamus, Wikipedia).
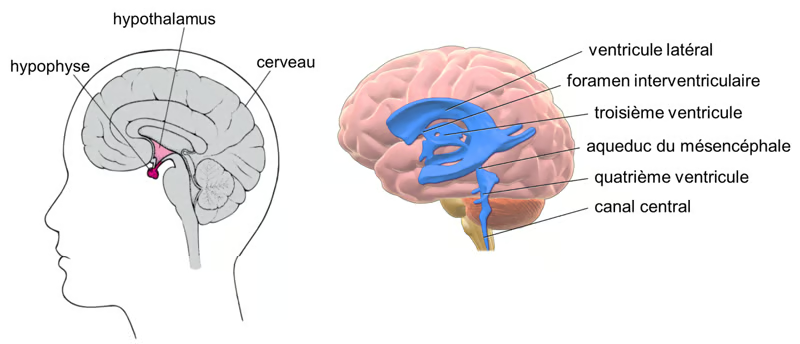
Figure. Hypothalamus and cerebrospinal fluid. On the left, the position of the hypothalamus and pituitary gland in the brain. On the right, the various compartments/ventricles containing the cerebrospinal fluid. (Cerebrospinal fluid, Wikipedia). Sources : Manuel MCD et Bruce Blaus sous licence CC BY 3.0.
Tanycytes are specialized glial cells of the hypothalamus, located in the median eminence, which line the third ventricle (Mullier et al. J Comp Neurol 2010; Prevot et al. Handb Clin Neurol 2021; Elizondo-Vaga et al. J Cell Mol Med 2015; Rodriguez-Cortes et al. Trends Endocrinol Metab 2025; Balland et al. Cell Metab 2014; Imbernon et al. Cell Metab 2022; Fong et al. Front Neurosci 2023). Tanycytes possess long extensions that come into contact with the blood vessels of the pituitary portal system. Tanycytes thus act as a bidirectional bridge between the cerebrospinal fluid and the bloodstream, as shown in the figure below.
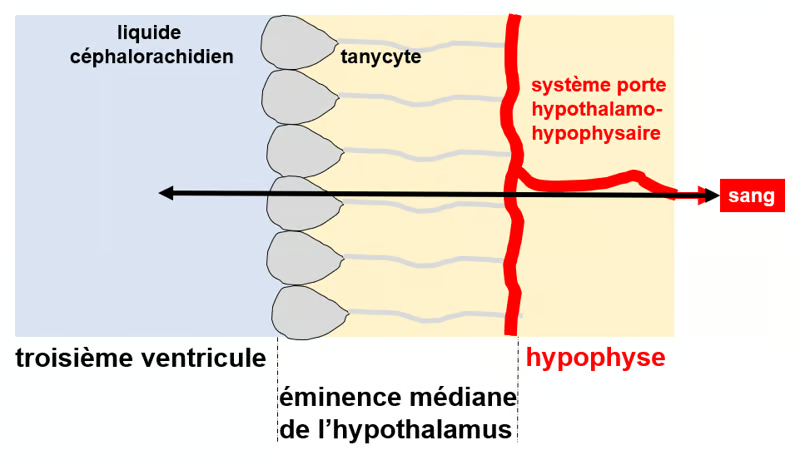
Figure. Tanycytes as a means of transport between the third ventricle and the peripheral blood. Tanycytes consist of a cell body (gray surrounded by black on the left) and long, thin processes (gray line). The cell bodies line the walls and floor of the third ventricle (light blue on the left), and the processes terminate in terminal feet in contact with the fenestrated capillaries (in red) of the hypothalamic-pituitary portal system. Tanycytes thus serve as a bridge between the cerebrospinal fluid and the peripheral blood circulation.
Results of this study (Sauvé et al. Cell Press Blue 2026)
Several figures from the article are presented here to support the authors' conclusions.
- Experiments on rodent cell cultures and animal models have shown that tanycytes transport Tau protein from the cerebrospinal fluid (CSF) into the blood.
- Blocking this transport (through appropriate experimental procedures) reduces this clearance, leads to an accumulation of Tau protein in the CSF, and accelerates/worsens the pathology.
- The study also examined Tau protein levels in the body fluids of patients. In individuals with Alzheimer's disease, the plasma/CSF ratios of total and phosphorylated Tau proteins were decreased, consistent with impaired clearance of Tau protein from the brain into the bloodstream.
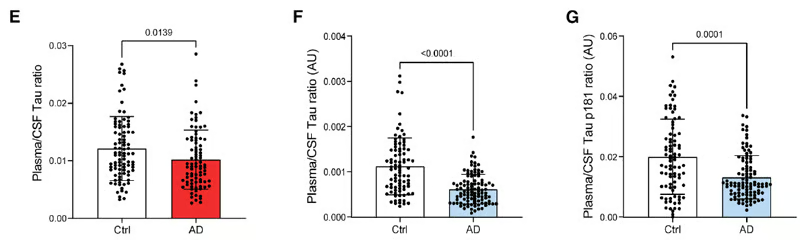
Figure 3 (E, F, G) from the article by Sauvé et al. 2026. (E): Ratio of plasma (blood)/CSF levels of Tau protein in controls (Ctrl) and patients with Alzheimer's disease (AD), extracted from the ADNI database (controls: n = 91; AD: n = 86). Data were analyzed using a two-tailed Mann-Whitney U test (U = 3077, p = 0.0139). Results are presented as means ± standard deviations. (F) Ratio of plasma (blood)/CSF levels of Tau protein in controls and patients with Alzheimer's disease (AD), extracted from the BioCogBank cohort (controls: n = 86; AD: n = 104). Data were analyzed using a two-tailed Mann-Whitney U test (U = 2118, p < 0.0001). Results are presented as means ± standard deviations. (G) Ratio of plasma (blood)/CSF levels of hyperphosphorylated Tau-181 protein in controls and patients with AD, extracted from the BioCogBank cohort (controls: n = 86; AD: n = 104). Data were analyzed using a two-tailed Mann-Whitney U test (U = 3051, p < 0.0001). Data are presented as means ± standard deviations.
- In post-mortem brain tissue from Alzheimer's patients, tanycytes contain pathological Tau protein but exhibit significant degeneration: their processes are fragmented/disorganized. Consequently, their Tau protein transport activity is greatly reduced.
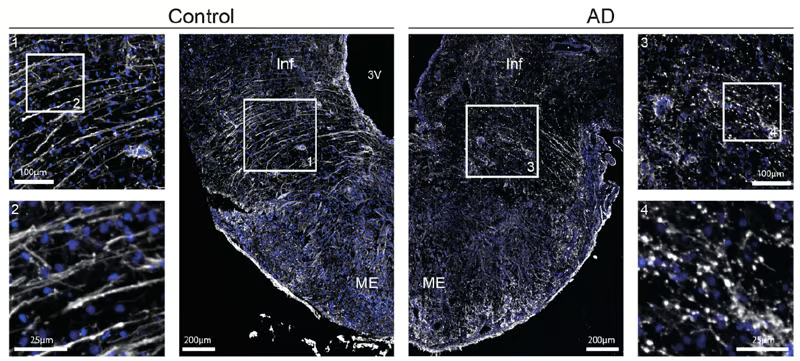
Figure 5 (A) from the article by Sauvé et al. 2026. Photomicrographs of tanycytes in the median eminence of the hypothalamus from controls (left, Control) and AD patients (right, AD), labeled by immunofluorescence for vimentin (white). Insets: higher-magnification images of tanycytic processes in controls (inset 2, bottom left) and AD patients (inset 4, bottom right), showing process fragmentation in the latter. Vimentin (appearing white), used as a marker in these micrographs, is a protein expressed in tanycytes.
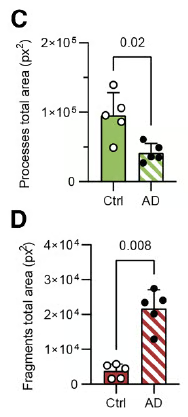
Figure 5 (C, D) from the article by Sauvé et al. 2026. (C) Graphique représentant la surface totale couverte par les prolongements des tanycytes chez les témoins (Ctrl, n = 5) et les patients Alzheimer (AD, n = 5). Un test de Mann-Whitney bilatéral a été utilisé (U = 1, p = 0,02). Les données sont présentées sous forme de moyennes ± écart-type. (D) Graphique représentant la surface totale couverte par les fragments de prolongements de tanycytes chez les témoins (Ctrl, n = 5) et les patients Alzheimer (AD, n = 5). Un test de Mann-Whitney bilatéral a été utilisé (U = 0, p = 0,008). Les données sont présentées sous forme de moyennes ± écart-type.
- The body of animal, cellular, and human data supports the existence of a "tanycyte shuttle" between the brain and the blood, the dysfunction of which could accelerate the accumulation of Tau protein.
- The authors sought to understand why tanycytes are severely damaged and non-functional in the brains of Alzheimer's patients. To do so, they undertook a study of the transcriptome (the complete set of mRNAs) of these cells.
- Sequencing the tanycyte mRNAs revealed major alterations, including: i) underexpression and dysregulation of genes involved in vesicular transport, anchoring, and exocytosis (cell exit), ii) significant activation of various cellular stress response pathways, and iii) ultimately, the triggering of cell death mechanisms.
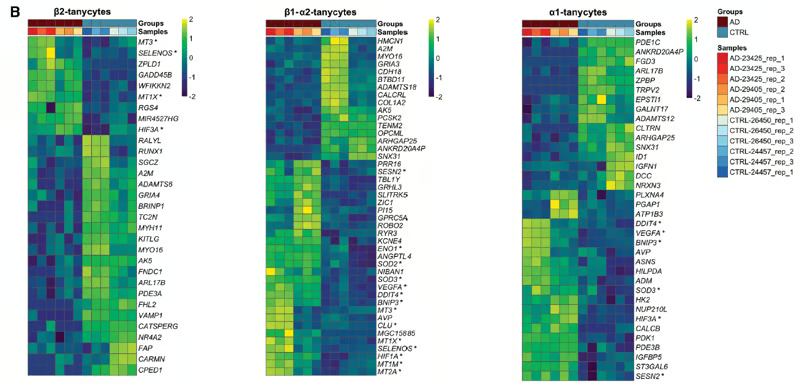
Figure 8 (B) from the article by Sauvé et al. 2026. Heat map showing normalized, row-scaled expression values of differentially expressed genes in β2, β1/α2, and α1 tanycytes. These different tanycytes are located on the third ventricle at different sites. (Fong et al. Front Neurosci 2023). The data were analyzed using a near-likelihood F-test (QLF) comparing aggregate counts per sample, according to the conditions. The significance level was set at a false positive rate (FDR) ≤ 0.05. This figure clearly shows that genes are expressed at very different levels between tanycytes from different Alzheimer's patients (brown bar at the top of the figure, AD) and those from control cases (CTRL); these genes are generally expressed at a higher level for yellow and a lower level for dark blue. Each row represents a different gene, the name of which is placed to the right of each frame.
- This degeneration impairs the cells' ability to clear Tau protein from the cerebrospinal fluid, leading to its pathogenic aggregation, spread, and accumulation in the brain, and contributing to the progression of Alzheimer's disease.
Study summary
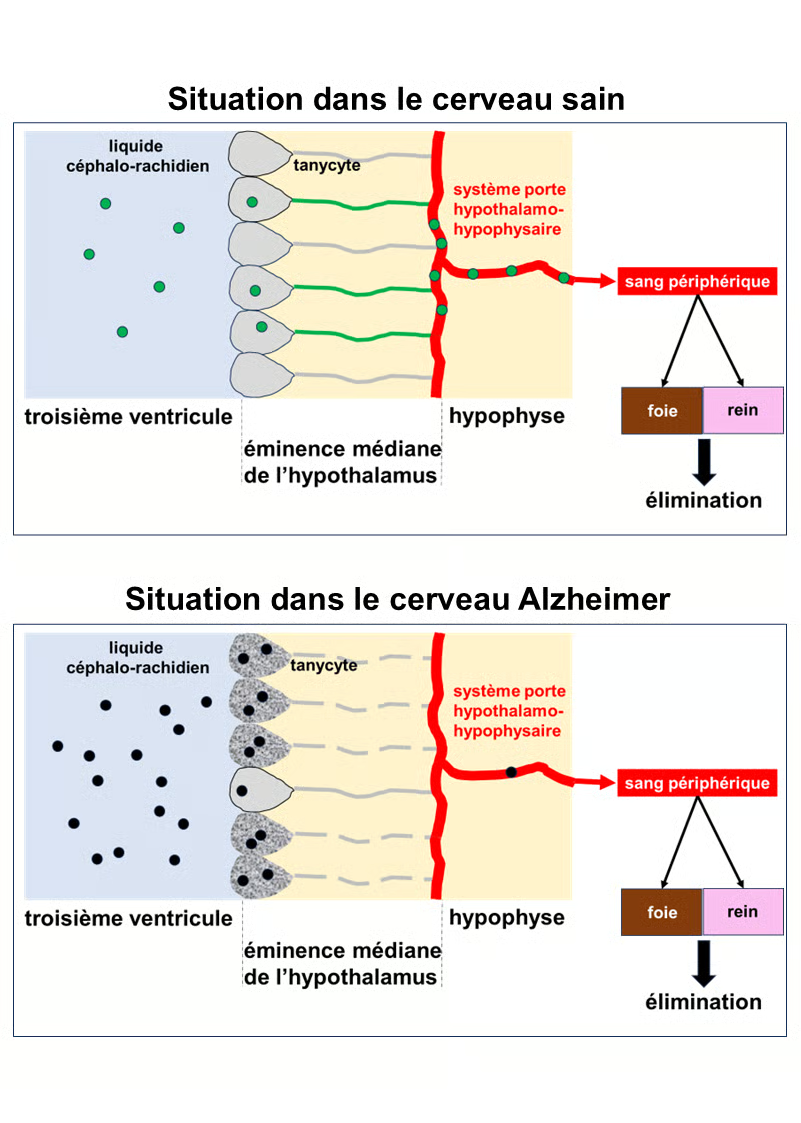
Top section: Situation in the healthy brain. Normal Tau protein, represented by a green circle, is present in the cerebrospinal fluid (light blue). It is taken up by the tanycytes of the hypothalamus, which line the third ventricle of the brain. The tanycytes transport the Tau protein into the bloodstream via the pituitary portal system. This protein is then eliminated from the body by the liver and kidneys.
Bottom section: Situation in the Alzheimer's brain. The Tau protein is abnormal and toxic (hyperphosphorylated). It is represented by the black circles. In the Alzheimer's brain, the tanycytes are degraded, and their extensions, which connect to the pituitary portal system, are fragmented and therefore non-functional (gray dotted lines). They can incorporate the Tau protein, but cannot export it into the bloodstream. It accumulates in the cerebrospinal fluid and the brain, continuing its toxic action on neurons.
Authors' conclusion
This study highlights the function of tanycytes as a potential new therapeutic target in the treatment of Alzheimer's disease. Preserving or restoring their integrity could improve the clearance of the Tau protein, slow the progression of the disease, and, beyond that, limit other pathologies (dementias) linked to the Tau protein. The researchers emphasize that maintaining tanycyte function is probably not the only solution to Alzheimer's disease, but could represent a promising new avenue. This study refocuses attention on this still poorly understood hypothalamic cell type as a key player in the pathogenesis of Alzheimer's disease, beyond traditional neuronal or amyloid perspectives.
Beyond the involvement of tanycytes in Alzheimer's disease, this study shows that these cells play an important role in other functions, including their presumed response to various stressors, their better-known roles as transporters of metabolic signals, and their role as regulators of hypothalamic hormone secretion.
Further studies should shed light not only on this new role in cerebral elimination mechanisms, but also on the pathophysiology of neurodegenerative diseases.
To be continued...